[ad_1]
Instead of storing electrons within the structure of a battery, a flow battery stores electrons in the form of chemicals in tanks, which are pumped into an electrolytic cell when charge or discharge is required.
The action decouples power and energy storage, which are inextricably linked in a conventional battery: in a flow battery, energy storage capacity is proportional to the size of the liquid storage tanks, while power density is proportional to the size of the electrolytic cell and capacity of the pumps.
A potential application for flow batteries is buffering intermittent renewable power sources to power grids.
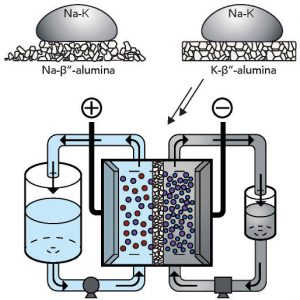
Key to using Na-K is the development by Stanford of a suitable solid electrolyte separator membrane for the electrolytic cell, that has to be an electrical insulator while passing metal ions.
The team came up with K-β″-alumina (‘potassium beta prime prime alumina’), a form of Al2O3 complexed with potassium ions that blocks electrons while passing potassium ions.
 PhD student team members (from the left) Geoff McConohy, Antonio Baclig and Andrey Poletayev
PhD student team members (from the left) Geoff McConohy, Antonio Baclig and Andrey Poletayev
The material is a spin on Na-β″-alumina, a sodium ion conductor used in high-temperature sodium-sulphur batteries.
On the other side of the cell, they have tried a number of aqueous and non-aqueous solutions that can accept K+ ions – there is a substantial list of them in ‘High-Voltage, Room-Temperature Liquid Metal Flow Battery Enabled by Na-K|K-β″-Alumina Stability‘, a paper published abut the research in Joule.
Results are mixed.
This type of membrane is most effective at temperatures higher than 200°C, according to the University, but a thinner membrane boosted room-temperature output. It did block both electrons and sodium ions, as it should, but quickly degraded with water-based positive electrodes, although non-water-based options showed promise.
Output is around 3.1-3.4V, with current limited by the membrane. Power, according to the paper, is up to 65mW/cm2 at 22°C and >100mW/cm2 at 57°C.
“We still have a lot of work to do. A new battery technology has so many different performance metrics to meet: cost, efficiency, size, lifetime and safety,” said Baclig. “We think this sort of technology has the possibility to meet them all, and using Earth-abundant materials, which is why we are excited about it.”
This project was funded by Stanford’s TomKat Center for Sustainable Energy, the Anthropocene Institute, the State Grid Corporation of China through Stanford’s Energy 3.0 corporate affiliate program, the National Research Foundation of Korea, the US National Science Foundation and Stanford Graduate Fellowships.
[ad_2]
Source link