[ad_1]
“What we have shown here is that we can make tiny, conductive wires of the smallest possible size that essentially assemble themselves,” says Stanford’s Hao Yan, “the process is a simple, one-pot synthesis. You dump the ingredients together and you can get results in half an hour. It’s almost as if the diamondoids know where they want to go.”
Although there are other ways to get materials to self-assemble, this is the first one shown to make a nanowire with a solid, crystalline core that has good electronic properties.
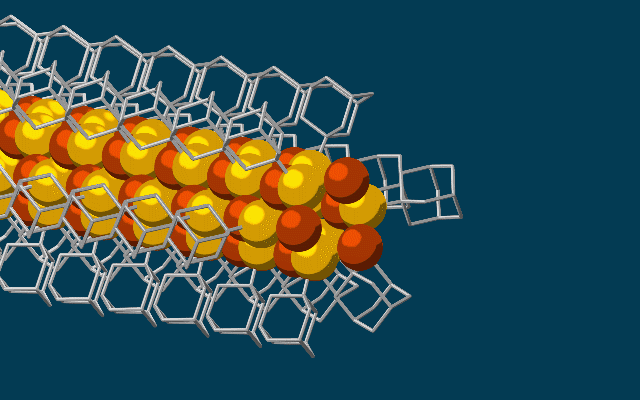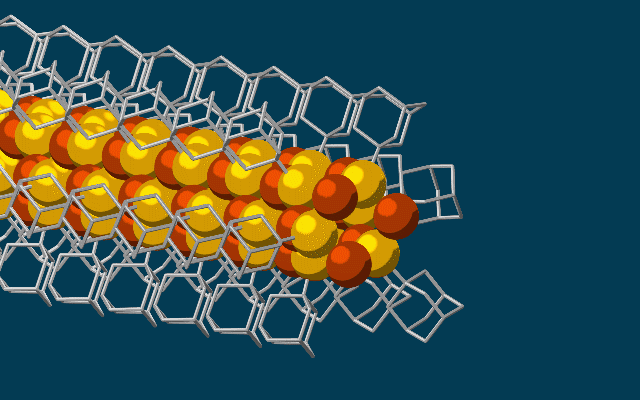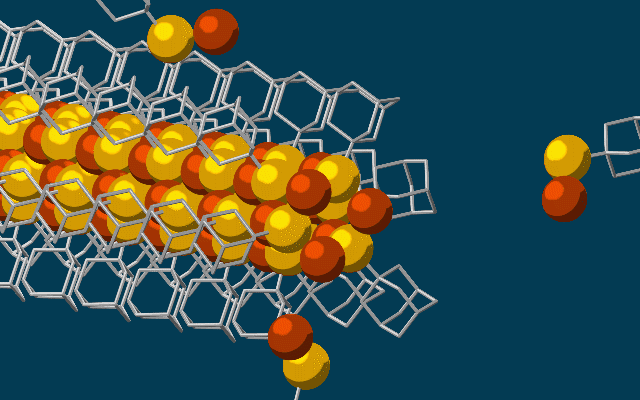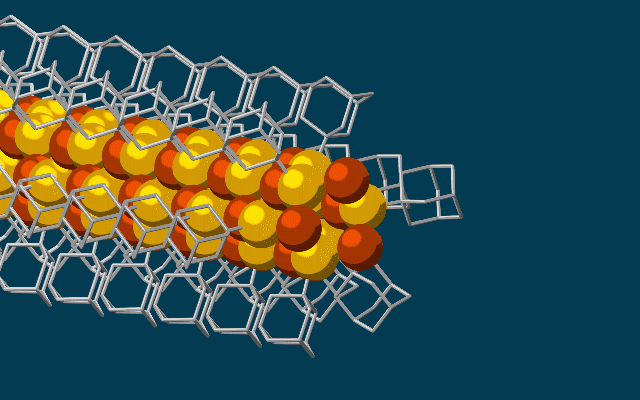
The needle-like wires have a chalcogenide semiconducting core surrounded by the attached diamondoids which form an insulating shell.
The method allows researchers to assemble those materials with atom-by-atom precision and control.
The diamondoids are found naturally in petroleum fluids and are extracted and separated by size and geometry in a SLAC laboratory.
For this study, the research team took advantage of the fact that diamondoids are strongly attracted to each other, through van der Waals forces.
They started with the smallest possible diamondoids – single cages that contain just 10 carbon atoms – and attached a sulfur atom to each. Floating in a solution, each sulfur atom bonded with a single copper ion. This created the basic nanowire building block.
The building blocks then drifted toward each other, drawn by the van der Waals attraction between the diamondoids, and attached to the growing tip of the nanowire.
The team has already used diamondoids to make one-dimensional nanowires based on cadmium, zinc, iron and silver, including some that grew long enough to see without a microscope, and they have experimented with carrying out the reactions in different solvents and with other types of rigid, cage-like molecules, such as carboranes
[ad_2]
Source link