[ad_1]
“Pseudocapacitance is a property of polymer and composite supercapacitors that allows ions to enter inside the material and thus pack much more charge than carbon ones that mostly store the charge as concentrated ions – in the so-called double layer – near the surface”, said QMUL.
According to the researchers, conducting polymers have high theoretical specific capacitance, but poor ion mobility means only the top few nanometres of such materials are ever used.
Making the conducting polymer with a nano-porous structure can turn more of it into available surface “but this can be expensive, hard to scale up, and often results in poor mechanical stability”, said QMUL.
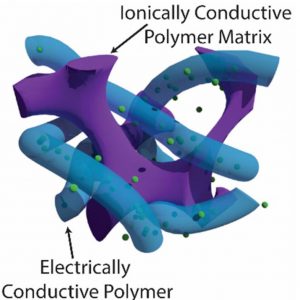
However, by creating a itimate tangle, called a semi-interpenetrating network (sIPN), of an electrically-conductive (pseudocapacitive) polymer in an ionically-conductive polymer matrix, virtually all of the pseudocapacitive polymer becomes available.
According to the paper ‘Semi-interpenetrating polymer networks for enhanced supercapacitor electrodes‘ in ACS Energy Letters, a free-standing PEDOT/PEO (3,4-ethylenedioxythiophene)/poly(ethylene oxide)) sIPN film yields 182F/g specific capacitance, 97.5% capacitance retention after 3,000 cycles, and such pysical flexibility that the electrodes can be bent to <200μm radius without breaking.
“Our supercapacitors can store a lot of charge very quickly, because the thin active material is always in contact with a second polymer which contains ions,” said project leader Stoyan Smoukov. “This interpenetrating structure enables the material to bend more easily, as well as swell and shrink without cracking, leading to greater longevity.”
The team is now designing and evaluating a range of materials for interpenetrating polymer supercapacitors.
[ad_2]
Source link