[ad_1]
The aim is to allow disposable rather than rechargeable cells to be used – to remove the need for charging components and, in clinical environments, remove the need for contacts, clips, and charging ports where germs may linger, said the firm, adding: “In clinical environments, primary cell architectures can create hermetically sealed units to safely disinfect between use or even dispose of completely to inhibit patient-to-patient infection.”
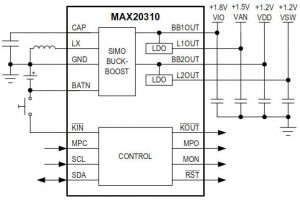
Using a single-inductor two-output architecture with two linear regulators, the chip drives four power outputs from a single inductor.
For pre-use product storage, there is a 10nA ‘battery seal’ mode.
Operation is over -40 to +85degC, and it comes in a 1.63 x 1.63mm wafer-level package.
An application circuit in the abridged data sheed (full not publicly available) shows the chip being used with a MAX32620 ARM Cortex-M4F based microcontroller.
There is an evaluation kit: MAX20310EVKIT#
Dual-output buck-boost
- Vin 0.7-2.0V (250mW max)
- Vout 0.9-4.05V
- 1μA quiescent
- 84% efficient for 1.8Vout 10mA
Dual LDOs
- Supplied from buck-boost
- Inputs Supplied by Dual Buck-Boost Outputs
- Vout 0.5-3.65V
- Iout max 50mA
- 0.5μA quiescent
- Configurable as load switch
[ad_2]
Source link